Published September 11,2023
Subscribe
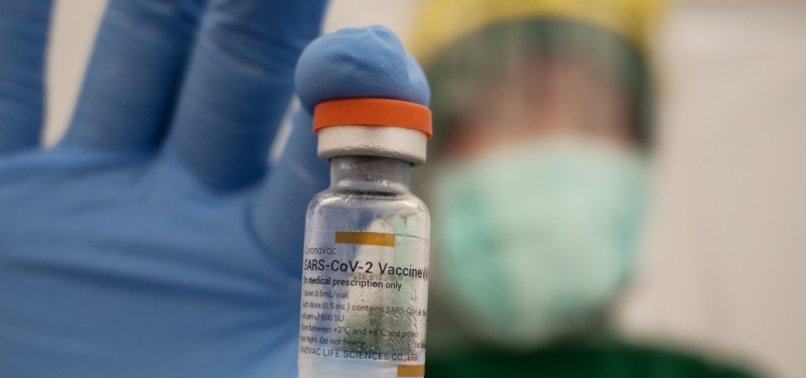
The Chinese biotech agency, Sinovac Biotech, mentioned on Monday that it had began a Phase 2 trial of its SA55 injection, which it hopes will probably be permitted as a Covid-19 therapy.
The Phase 1 examine, which was accomplished lately, had confirmed the preliminary security profile of SA55 in 40 wholesome adults aged 18 to 65 in China.
The Phase 2 examine will consider the flexibility of SA55 to scale back SARS-CoV-2 viral load and alleviate scientific signs in almost 150 sufferers to be enrolled with gentle to average Covid-19.
Source: www.anews.com.tr